The app for independent voices
🔥A New Diagnosis Code for COVID-19 Vaccine Adverse Effects Needs Your Support: CDC’s New ICD-10 Code T50.B25 Open for Comment — a Message from React19 (posted 03/26/26)
Dear Readers,
React19 just issued an important call to action regarding a new code to indicate COVID shot injury. This code both recognizes the injury and makes it easier for those suffering from COVID shot injuries to receive treatment.
We’ve copied the message below, with minor changes for Substack. Images from article:
__________
How to Submit Your Comment
Takes less than five minutes. Every comment strengthens the case for adoption.
Deadline: Friday, May 15, 2026
Send comment to: mailto:nchsicd10cm@cdc.gov
1. Copy template email below. Subject line and body are ready to use.
2. Personalize it. Fill in your name, title, organization, city, state, and contact information at the bottom. If you are a clinician, coder, researcher, or patient advocate, say so — it adds weight to your comment.
3. Send to mailto:nchsicd10cm@cdc.gov by Friday, May 15, 2026. No attachments necessary.
4. Share this email. Forward to colleagues, patient communities, and anyone who understands the importance of accurate medical documentation.
______
Template Email
TO:mailto:nchsicd10cm@cdc.gov
SUBJECT: Public Comment in Support of Proposed ICD-10-CM Code T50.B25 – Adverse Effect of COVID-19 Vaccines
Dear NCHS ICD-10-CM Coordination and Maintenance Committee,
I respectfully write in support, and urge the Committee to adopt code T50.B25, including its three encounter-specific variations:
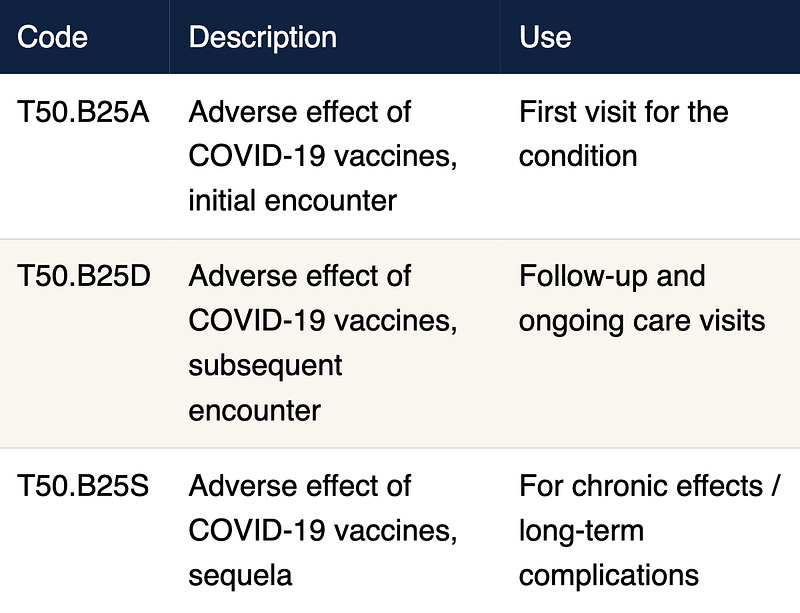
T50.B25A – Adverse effect of COVID-19 vaccines, initial encounter
T50.B25D – Adverse effect of COVID-19 vaccines, subsequent encounter
T50.B25S – Adverse effect of COVID-19 vaccines, sequela
Currently, no specific ICD-10-CM code exists for adverse effects following COVID-19 vaccination. In practice, a lack of this code has resulted in miscoding. This miscoding obscures the true nature of these cases in clinical records, research datasets, and public health surveillance systems, and conflates distinct patient populations.
A dedicated code under the new subcategory T50.B2 would allow clinicians, researchers, and public health professionals to:
Accurately document adverse effects specific to COVID-19 vaccines
Support ongoing research and surveillance of post-COVID-19 vaccine conditions
Improve the patients' ability to receive care.
Medical coding is fundamental to the integrity of our health data infrastructure. The adoption of T50.B25 represents a straightforward and meaningful step toward ensuring that patients experiencing adverse effects following COVID-19 vaccination receive accurate and complete documentation of their conditions, separate and distinct from those who experienced adverse effects from COVID-19 infection itself.
Thank you for your consideration of this proposal and for your continued work on the ICD-10-CM code set.
Respectfully,
[Your Full Name]
[Title / Organization] - optional
[City, State] - optional
[Email Address]
_____
Copy, personalize, and send to mailto:nchsicd10cm@cdc.gov by May 15, 2026.
_____
Background & Proposal
U.S. Has No Specific Diagnosis Code for COVID-19 Vaccine Adverse Effects — A Proposed Fix Is Now Before the CDC
For the first time, the federal agency that maintains the nation's medical coding system has proposed a dedicated code for adverse effects of COVID-19 vaccines. What it means, why it matters, and what happens next.
_____
More than four years into the rollout of COVID-19 vaccines, the United States still has no specific diagnosis code to document adverse health conditions that arise from them. That may be about to change. At the March 17–18, 2026 ICD-10 Coordination and Maintenance Committee Meeting, the National Center for Health Statistics (NCHS) presented a formal proposal to add T50.B25x Adverse Effect of COVID-19 Vaccines, to the ICD-10-CM code set used by every U.S. healthcare provider, hospital, insurer, and public health agency.
The proposal was in response to a request from React19, a nonprofit organization representing individuals experiencing persistent health conditions following COVID-19 vaccination. Public comment period is now open; comments must be submitted to NCHS by Friday, May 15, 2026.
What did REACT19 present to CDC?
_____
What Is ICD-10-CM and Why Does It Matter?
International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) is the standardized system U.S. providers use to classify and document diagnoses. These codes drive everything from insurance reimbursement and hospital resource allocation to public health surveillance and clinical research. When a condition lacks its own code, it becomes invisible, providers have no reliable way to document it, researchers have no way to count it, and policymakers have no data to act on.
The importance of dedicated codes was demonstrated clearly with the October 2021 introduction of U09.9 (Post-COVID-19 condition, unspecified) for Long COVID. That single code enabled systematic documentation of Long COVID cases across the country, supported surveillance, and helped launch a wave of clinical and epidemiological research, even before the full biology of the condition was understood.
_____
Problem: No Code, No Data
Currently, when a patient presents with an adverse effect following COVID-19 vaccination, their provider has no specific code to assign. NCHS guidance technically points to T50.B95- (Adverse Effect of Other Viral Vaccines) as a catch-all, but this nonspecific code has caused substantial confusion for providers, citing that COVID vaccines are not typically viral, but rather mRNA.
"In practice, the absence of a specific code has led to widespread miscoding of COVID-19 vaccine adverse effects under U09.9 Long COVID, conflating two distinct patient populations and corrupting the data for both." - Dr Joel Wallskog
This is not a minor administrative inconvenience. Downstream consequences are significant: patients are misdocumented in their medical records, the true incidence of post-COVID vaccination adverse conditions is unknown, research cohorts are contaminated with misclassified patients, and individuals seeking disability benefits or continuity of care face documentation gaps that undermine their cases.
_____
What the Proposal Creates
REACT19 proposed a new sub-subcategory, T50.B and 3 new codes:
These three variations are critical. The inclusion of a sequela designation (T50.B25S) is particularly significant for patients experiencing prolonged or chronic post-vaccination conditions, as it provides a mechanism to document the ongoing relationship between a patient's current symptoms and their prior vaccination — something the current nonspecific codes cannot accomplish.
When used correctly, T50.B25 would be assigned as a secondary code alongside specific manifestation codes describing patient's actual symptoms or conditions. For example:
_____
Example: Clinical Coding Scenario
Primary
T50.B25S — Adverse effect of COVID-19 vaccines, sequela
_____
Secondary
G90.1 — Postural Orthostatic Tachycardia Syndrome (POTS)
_____
Secondary
G62.81 — Small fiber neuropathy
_____
Secondary
R53.82 — Chronic fatigue
_____
Secondary
D89.40 — Mast cell activation syndrome (MCAS)
_____
Clinical Landscape: A Multisystem Condition
The population that stands to benefit from this code includes individuals experiencing persistent, multisystem symptoms following COVID-19 vaccination — a presentation sometimes referred to as Post-COVID-19 Vaccination Syndrome (PCVS). Published research has documented involvement across neurological, autonomic, cardiovascular, musculoskeletal, pulmonary, gastrointestinal, immunological, hematologic, and constitutional systems. Conditions documented in the peer-reviewed literature include POTS, small fiber neuropathy, myocarditis, mast cell activation syndrome, and chronic fatigue, among others.
There is meaningful clinical overlap between PCVS and Long COVID, and research is actively exploring shared and divergent pathophysiological pathways. Precisely because of this overlap, clean coding infrastructure is essential, without a distinct code for vaccine adverse effects, cases of PCVS and Long COVID will continue to be conflated in the data, hampering both populations' access to research, recognition, and care.
_____
What Happens After Comment Period
ICD-10 Coordination and Maintenance Committee reviews all public comments submitted before May 15th, 2026. Comments from clinicians, coders, researchers, public health professionals, and affected individuals all carry weight. Following the review period, Committee will issue a final determination. If adopted, new ICD-10-CM codes typically take effect at the start of the next fiscal year coding cycle.
This is a rare and direct opportunity for the public, including patients and providers, to formally influence the federal coding infrastructure that shapes medical documentation, research, and policy. The window is short. Comments must be received at mailto:nchsicd10cm@cdc.gov by Friday, May 15, 2026.
"Without proper diagnosis codes, these emerging complex chronic illnesses remain invisible to surveillance systems, preventing early recognition, appropriate treatment, and proper resource allocation." - Brianne Dressen
_____
The proposed code T50.B25 is not a political statement. It is a data infrastructure decision. Accurate coding does not create conditions, it makes existing ones visible.
The case for adoption is straightforward: providers need a specific code to document these cases correctly, researchers need clean data to study them, and patients deserve medical records that accurately reflect what happened to them.
You can review the full CDC proposal here: (p 16-17): cdc.gov/nchs/data/icd/t…