Make money doing the work you believe in
So, Odyssey Therapeutics is pricing its IPO targeting roughly $238 million in proceeds and an ~$810 million valuation. J.P. Morgan, TD Cowen and Cantor are among underwriters.
The timing is probably right, as Kailera just had one of the biggest biotech IPOs on record. April was the strongest month for new filings in over four years, according to Renaissance Capital.
What makes this story interesting is not even Odyssey's IPO progress per se, but the therapeutic thesis behind it that they use to go public.
Their lead asset, OD-001 (OD-07656 in the S-1), is an oral small molecule RIPK2 scaffolding inhibitor entering Phase 2a for ulcerative colitis and Crohn's disease.
The angle here is that virtually every approved IBD therapy like TNF blockers, IL-23 inhibitors, integrin blockers, JAK inhibitors, targets the adaptive immune system. Remission rates remain modest: roughly 20-30% of patients respond to any given treatment (placebo-adjusted), and up to 45% of initial responders lose response over time.
Odyssey's argument is that the problem starts upstream, in the innate immune system. RIPK2 sits at a signaling node where bacterial byproducts activate innate immune cells, which then produce the cytokines that kick off the adaptive response everyone else is trying to block downstream.
Their preclinical data shows RIPK2 gene activation remains elevated in patients who fail standard-of-care therapies like infliximab, vedolizumab and ustekinumab, probably suggesting the innate pathway keeps firing even when you shut down adaptive targets.
They also have preclinical biopsy data showing OD-001 reduced expression of inflammatory and fibrotic gene signatures, which is notable because fibrosis in IBD drives surgical interventions and there's nothing approved that addresses it directly.
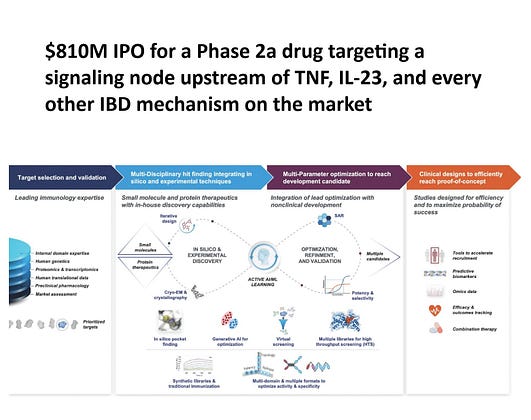
Beyond RIPK2, the platform is worth a look.
They run three small molecule discovery engines — an AI/ML platform, a natural products library screening hundreds of thousands of microbial strains, and a covalent chemistry platform, alongside a VHH single-domain antibody platform for protein therapeutics. The Pfizer and J&J collaborations were built around these capabilities, though Odyssey notes in the filing that it doesn't expect additional revenue from either partnership.
A collaboration with Terray Therapeutics on IRF5 inhibition for autoimmune disease is ongoing.
Phase 2a topline induction data for the UC monotherapy trial is expected in the first half of 2026. That's the near-term catalyst that will determine whether the biology translates.
An $810 million valuation for a company with one clinical-stage asset and five preclinical programs in a crowded indication is not cheap... But if RIPK2 inhibition works in patients, the mechanistic differentiation from everything else in IBD is real.
Let me know if I missed any other similar work in IBD space or similar other things? Would be interestiung to know.