The app for independent voices
⚡Breaking: MODERNA and MERCK just reported a five year follow up in high risk resected #melanoma for their individualized #mRNA neoantigen therapy (V940, intismeran autogene).
In KEYNOTE-942 (phase 2b, n=157) in stage 3/4 melanoma after complete resection, V940 plus Keytruda reduced the risk of recurrence or death by 49% vs Keytruda alone at a median five years (HR 0.51, 95% CI 0.294-0.887). Same magnitude they reported at three years.
A few takeaways:
1. Durability is the whole point. Plenty of trials look good early but far fewer still look good at five years in the adjuvant setting. This one does.
2. Melanoma is an ideal proving ground, but the real prize is portability to colder, lower mutation tumors.
3. Execution risk is still the product. Phase 3 is fully enrolled. Now it’s turnaround time, manufacturing reliability, and consistency across key risk subgroups.
4. Commercial upside is massive, and payer scrutiny will be too if pricing is additive on top of Keytruda. Analysts are already floating Keytruda-like pricing and multibillion peak sales in melanoma.
For context, the American Cancer Society estimates about 112k new melanoma diagnoses in the US in 2026.
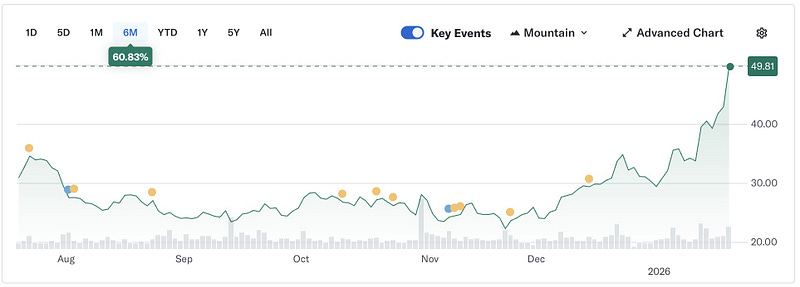
By late 2025, Moderna was the most shorted stock in the S&P 500 by percent of float, which tells you how skeptical the market was coming into 2026. This is also a strong way to start 2026 for Moderna. After the post Covid reset, they need durable, platform defining clinical wins that show mRNA can be a franchise beyond infectious disease. Five year durability in a flagship immuno-oncology combination is exactly that kind of signal.
I did a deep dive last year on where mRNA could go beyond COVID, including why personalized mRNA is a believable new franchise if they can execute at scale: