The app for independent voices
Is a “pill penalty” shaping the future of post-approval cancer research? Early data suggest the Inflation Reduction Act may be disincentivizing trials for small molecule drugs.
---
Under the IRA, small molecule drugs become eligible for Medicare price negotiation 7 years after FDA approval, which is 4 years earlier than biologics.
This difference in timing earned the description of a "pill penalty."
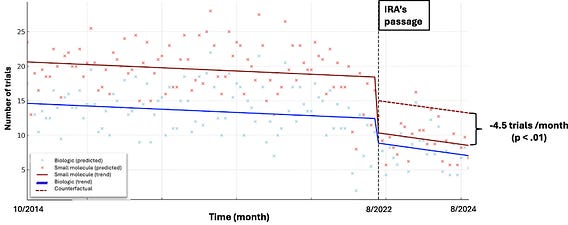
A recent difference-in-difference analysis of 4,367 post-approval phase I-III oncology trials (2014–2024) offers early evidence that the IRA’s #PillPenalty for price negotiation eligibility may be having real-world consequences.
---
The main results:
-A 45.3% drop in industry-sponsored post-approval trials for small molecules after the IRA, compared to 32.5% for biologics.
-Using biologics as a counterfactual, small molecules saw an additional decrease of 4.5 trials per month post-IRA.
The attached image shows that new trials dropped for both, but more for small molecules with the earlier negotiation eligibility.
---
In #oncology, two-thirds of industry-funded trials occur after FDA approval, and many small molecule drugs gain expanded indications years after initial launch.
Earlier price negotiation eligibility shortens the commercial window, reducing incentive to fund further research in other indications.
The study acknowledges limitations like potential unmeasured confounders and the challenge of isolating #policy effects in a post-COVID R&D landscape.
---
Evidence is accumulating that current IRA timelines unintentionally disfavor small molecule drugs.
A proposal to align the timelines for small molecules and biologics (to the longer timeline for biologics) nearly made it into the July 4 budget bill, but was ultimately removed before passage.
Should the form of a drug determine when it's eligible for price negotiation?